At times, potato growers may experience poor emergence of potato plants. Potato plants may not emerge properly for a number of reasons.
This publication is intended to provide a list of common problems that can cause poor potato emergence and stand. Utilizing this list can help growers more rapidly identify the cause and improve management of the crop and subsequent crops.
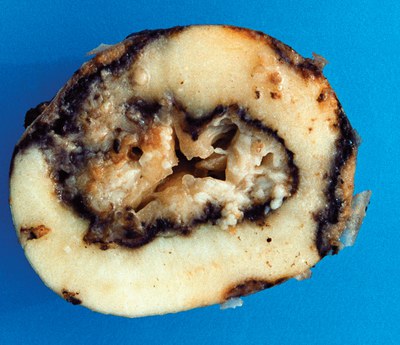
Planting healthy seed is one of the most important factors that allow growers to produce a high-quality and high-yielding potato crop. Certified seed should give optimum emergence when handled and suberized properly, especially keeping tubers free of bruises or injuries wounds. Optimum soil conditions will increase the probability of a successful plant stand.
Soil temperature and moisture are key factors in optimizing crop emergence. Ideally, the soil temperature and the seed temperature should be the same at planting; ideally 50 to 55 F (10 to 13 C). As soils warm, sprout growth becomes most rapid at 68 to 72 F (20 to 22 C).
Favorable soil moisture should be in the range of 75% to 85% available soil water. Seed treatments are available to reduce the incidence of fungal pathogens such as Fusarium, Helminthosporium and Rhizoctonia, but not for bacterial pathogens.
Slow emergence often results from planting in nonideal conditions or using lower-quality seed. But sometimes a poor stand follows high-quality seed and perfect planting conditions.
For example, planting good seed in a wet soil, either hot or cold, favors bacterial soft rot, which may result in poor stand. Or, planting good seed in ideal conditions, a poor stand can occur because of herbicide carryover in the soil or herbicide residues in the seed.
Potato emergence can be affected a number of ways. The following checklist is of common factors that occur before or after planting that reduce seed emergence.